September is Prostate Cancer Awareness Month, a dedicated time to recognize the brave individuals impacted by the disease and honor the ongoing research and innovation reshaping patient outcomes.
Nearly 314,000 patients in the United States will be diagnosed with prostate cancer this year, and the disease will claim approximately 35,770 lives, according to the American Cancer Society. In addition, the rate of men diagnosed1 with advanced disease has been growing in recent years.
What is Prostate Cancer?
Prostate cancer occurs when cells in the prostate gland start to grow abnormally. These abnormal cells can form tumors and spread to other parts of the body. Even though certain prostate cancers can grow rapidly, the majority advance slowly and respond well to treatment .
While anyone with a prostate may develop prostate cancer, some people face higher risk due to specific factors.2 African American men in the United States are 1.7 times more likely3 to develop prostate cancer compared to European American men. The disease occurs less often2 in Asian American, Hispanic, and Latino men. While rare in men younger than 40, the chance of having prostate cancer rises rapidly after 50, with about 6 in 10 prostate cancers2 found in men older than 65. Although the majority of cases have no family history, a man’s risk more than doubles2 if a close relative has had the disease.
Screening and Diagnosing Prostate Cancer
While symptoms of early prostate cancer are rare, they can include problems urinating and blood in the urine or semen. As the cancer advances and spreads, symptoms can include erectile dysfunction, pain in different parts of the body, weakness or numbness in the legs or feet, loss of bladder control, and fatigue.
To screen for prostate cancer, clinicians typically use a PSA (prostate-specific antigen) blood test, with or without a digital rectal exam (DRE). If cancer is suspected, additional testing is performed such as an MRI and prostate biopsy. The biopsy tissue is then graded using the Gleason score (Grade Group). Clinical stage is determined by combining PSA, biopsy results, and imaging findings. These imaging scans may also be used to evaluate whether the cancer has spread beyond the prostate (metastasis).
Learn more about when screening for prostate cancer is recommended.
The Challenge in Prostate Cancer Management
Prostate cancer is heterogeneous, meaning it doesn’t behave the same way in every person. Some prostate cancers grow very slowly, while others are aggressive and can spread quickly, making clinical decision-making challenging.
Historically, decisions were primarily made based on clinical and pathological factors, however, these factors alone do not always reflect the true biology of the tumor, which can lead to over- and under-treatment.
The good news is that gene expression testing is enabling clinicians and patients to better understand the unique biological characteristics of the disease and make more informed treatment decisions.
Genomic Technology as a Better Guide
Looking at the activity of certain genes in tumor cells using molecular tests like the Decipher Prostate Genomic Classifier can often better predict how likely a cancer will grow or spread.
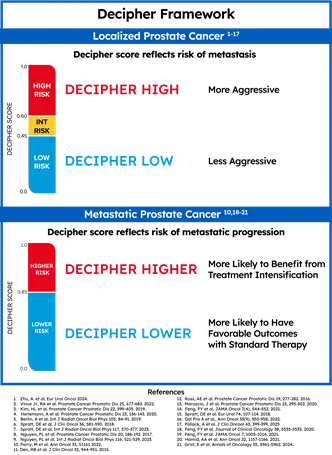
Decipher Prostate delivers trusted genomic insights to help clinicians and patients make confident, personalized treatment decisions across the prostate cancer continuum. The test is a highly accurate predictor of metastasis, prostate cancer-specific mortality, and overall survival in localized patients, among other endpoints. Additionally, for patients with metastatic prostate cancer, the Decipher score indicates the likelihood of cancer progression and survival benefit with treatment intensification.
What is the Decipher Prostate Test?
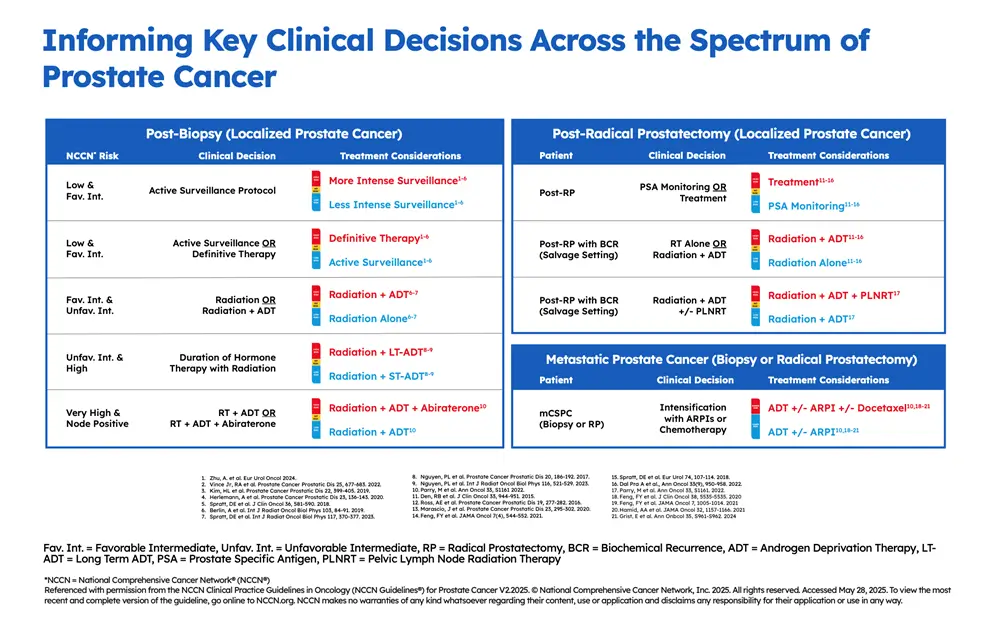
Decipher Prostate is a 22-gene genomic test that looks at the molecular characteristics of the prostate cancer tumor to assess its aggressiveness, or likelihood of spreading outside the prostate, providing actionable information for treatment decision-making across the spectrum of prostate cancer – post-biopsy, post-RP (radical prostatectomy), and advanced disease. It is performed on tissue collected during a patient’s existing prostate tumor biopsy or radical prostatectomy.
The result is a number from 0-1 (the patient’s Decipher score) classified as Decipher Low Risk, Decipher Intermediate Risk, or Decipher High Risk. For patients with localized or regional prostate cancer, a Decipher score indicates a patient’s risk of metastasis, helping to determine treatment timing and intensity. For patients with metastatic prostate cancer, the Decipher score indicates the likelihood of cancer progression and survival benefit with treatment intensification.
Patient Journeys: A Urologist’s Experience
Urologist Dr. Arnold Bullock, from the Washington University School of Medicine, shares his very personal story of being diagnosed with prostate cancer and using the Decipher Prostate test in his clinical practice for his patients. Watch his conversation with urologist and former colleague, Dr. Eric Kim, a key opinion leader from the University of Nevada, Reno School of Medicine.
Building Clinical Value and Rigorous Evidence
Decipher Prostate is the most extensively validated gene expression test in prostate cancer, with evidence from over 200,000 patients, more than 25 prospective randomized clinical trials, and upwards of 95 peer-reviewed publications demonstrating its clinical validity and utility.
It is the only gene expression test to achieve Simon Level I evidence status and inclusion in the risk-stratification table in the most recent version of the NCCN Clinical Practice Guidelines In Oncology (NCCN Guidelines®)4 for Prostate Cancer.
Recent Publication Highlight
Findings suggest Decipher Prostate can help clinicians better personalize chemotherapy decisions for patients with metastatic prostate cancer New data published online in Cell, from the randomized, prospective, Phase 3 STAMPEDE trial,5 show that Decipher Prostate predicts which patients with metastatic cancer are likely to benefit from treatment intensification with the chemotherapy docetaxel and which are not likely to benefit and can therefore avoid its unnecessary toxicity. Read Press Release.
What’s Next: Personalized Prostate Cancer Treatment Planning
As of June 2025, Decipher Prostate expanded into metastatic prostate cancer with the Decipher Prostate Metastatic Genomic Classifier. Approximately 10% (or about 30,000)1 of all prostate cancers diagnosed annually in the United States are metastatic, and Veracyte believes this important new tool will help clinicians make better-informed treatment recommendations.
Today, Veracyte estimates1 that about 40% of patients in the US with prostate cancer receive molecular testing to help guide their care, and as more patients embrace earlier testing, the opportunity for timely diagnosis can offer the potential to halt disease progression before it reaches advanced stages and improve long-term quality of life.
At the end of this month, multiple abstracts will be presented at ASTRO 2025 demonstrating Veracyte’s commitment to continue working with research collaborators to advance our collective knowledge of prostate cancer.
Throughout the prostate cancer care continuum, both clinicians and patients must navigate complex treatment planning decisions. Advanced genomic tests, like Decipher Prostate, capable of providing whole-transcriptome data and deeper molecular insights, will enable more precise, effective treatments for better patient outcomes.